Oxidation-reduction potential (ORP), commonly called Redox, is a measure of the tendency of a solution to either gain or lose electrons. Simply, it’s the measurement of a water body’s ability to cleanse itself or break down waste products, such as contaminants and organic materials. In the context of lakes and ponds, ORP is an important water quality parameter that can provide insight into the oxygen levels and other chemical reactions occurring in the water.
Oxidation-reduction potential for Lakes and Ponds
ORP is primarily determined by the balance between the amount of dissolved oxygen (a strong oxidizing agent) and other substances that can act as reducing agents, such as organic matter, hydrogen sulfide (H2S), and iron. When dissolved oxygen levels are high, the ORP is positive, indicating a strong oxidizing environment. Conversely, when oxygen levels are low, the ORP is negative, indicating a reducing environment. An environment where little to no oxygen is considered “reducing,” the opposite of an oxidative or oxidizing environment.

In lakes and ponds, ORP can be used to monitor changes in water quality over time, as well as to assess the effectiveness of management strategies such as aeration or chemical treatments. For example, adding oxygen to the water through nanobubble technology can increase the ORP and help maintain a healthy, oxygen-rich environment for aquatic life. Conversely, high levels of organic matter or other reducing agents can cause the ORP to drop, potentially leading to the accumulation of harmful compounds such as hydrogen sulfide.
Overall, ORP is an important tool for understanding the chemical dynamics of aquatic systems and can help guide management decisions aimed at promoting the health and sustainability of lakes and ponds.
How Can I Measure THE oxidation reduction potential for My Lake or Pond?
ORP is typically measured using a specialized instrument called an ORP meter, which consists of a probe and a display unit. The probe is inserted into the water and contains a sensing element made of a material such as platinum or gold that reacts to changes in the oxidation-reduction potential of the water.
When the probe is inserted into the water, a small electric current is applied to the sensing element, creating a voltage that is proportional to the ORP of the water. This voltage is then transmitted to the display unit, which shows the ORP reading in millivolts (mV).
To obtain accurate ORP measurements, it is important to calibrate the ORP meter periodically using standard solutions with known ORP values. Additionally, it is important to ensure that the probe is clean and free from any contaminants that could interfere with the measurement.

ORP measurements can be taken at different depths and locations within a lake or pond to obtain a more comprehensive understanding of the chemical dynamics and water quality conditions. However, it is important to note that ORP is just one of many factors that contribute to overall water quality and should be used in conjunction with other measurements and observations to inform lake and pond management decisions.
What Factors Affect My Lake’s ORP?
Understanding the factors that affect ORP can be useful in developing management strategies for lakes and ponds, such as aeration or chemical treatments. By monitoring ORP levels and identifying the underlying causes of any changes, lake and pond managers can make informed decisions to promote a healthy and sustainable aquatic ecosystem.
Several factors can affect ORP in lakes and ponds, including:
- Dissolved oxygen levels: As mentioned above, dissolved oxygen is a strong oxidizing agent and can increase the ORP of the water. Conversely, low oxygen levels can decrease the ORP and create a reducing environment.
- Organic matter: Organic matter can act as a reducing agent and lower the ORP of the water. This can occur naturally as a result of the decomposition of plant and animal material or can be exacerbated by excess nutrient inputs from sources such as fertilizer runoff or sewage.
- Temperature: Temperature can affect the rate of chemical reactions in the water, which can in turn affect the ORP. In general, warmer water tends to have a lower ORP than colder water.
- pH: The pH of the water can also influence the ORP. In acidic conditions, the ORP tends to be lower, while in alkaline conditions, the ORP tends to be higher.
- Redox-active substances: Certain substances in the water, such as iron or hydrogen sulfide, can act as reducing agents and lower the ORP.
Best Practices for ORP Management in Lakes and Ponds
Now that you know the importance of ORP for your lake’s health, here are several best practices for managing ORP in lakes and ponds:
Monitor ORP levels regularly
Regular monitoring of ORP levels can help identify changes in water quality and provide early warning of potential issues. ORP levels can be measured using a specialized instrument called an ORP meter.
Address the underlying causes of low ORP
If ORP levels are consistently low, it is important to identify and address the underlying causes. This may involve reducing nutrient inputs, increasing aeration, adding nanobubble generators to efficiently increase dissolved oxygen and increase chemical-free oxidation, or addressing other water quality issues.
Consider increasing dissolved oxygen with nanobubbles, which deliver additional benefits
Enriching water with nanobubble generators helps increase dissolved oxygen levels in the water, which can in turn increase the ORP. This can be achieved using diffused air systems, fountains, or other types of aeration devices, though nanobubbles have been used increasingly in the lake and pond management industry to efficiently increase dissolved oxygen throughout the water column, even penetrating the sediment layer to help break down muck, and injecting nanobubbles that have a variety of benefits for lakes and ponds.
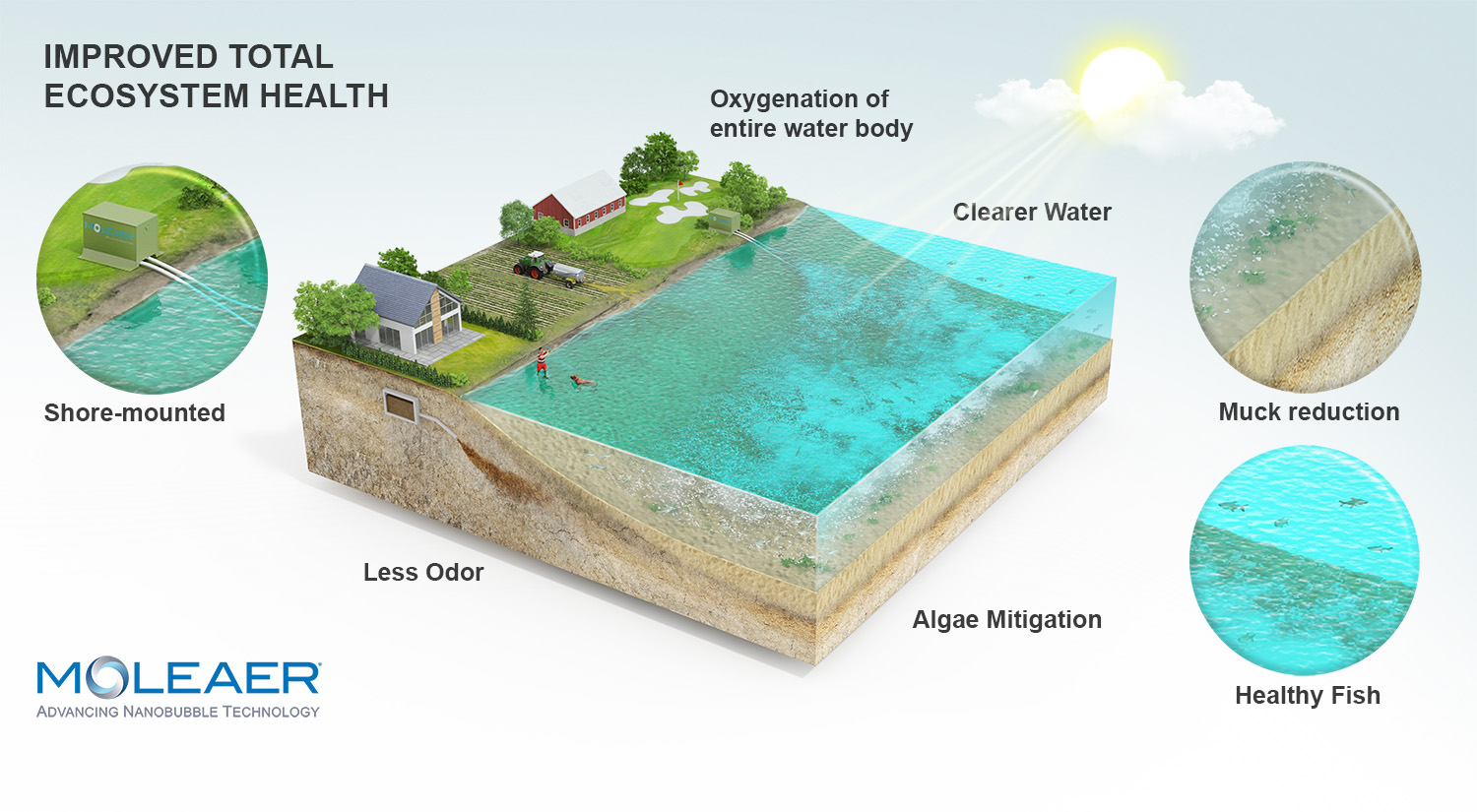
As nanobubbles are extremely small, stable and have a high surface area to volume ratio, they can remain in the water for a longer period of time than larger bubbles. This allows for a more efficient, stable transfer of oxygen into the water.
The increased dissolved oxygen levels from nanobubble technology and nanobubble's ability to enhance oxidation help create a more oxidative environment, raising ORP and promoting the growth of beneficial bacteria and other organisms that require oxygen. This also reduces the growth of harmful bacteria and algae that thrive in low-oxygen or reducing environments.
Moleaer offers two nanobubble generators specifically designed for lakes and ponds, allowing lake managers to harness the power of nanobubbles for chemical-free improvements. Depending on the size of the water body and goals, Moleaer’s team of experts will help you pick the best nanobubble generator for you. The Clear nanobubble generator is designed for larger lakes and ponds with flow rates of 50 or 150 GPM, while the Kingfisher is designed for smaller-sized lakes and ponds and has a 40 GPM flow rate. Both are easy to install and maintain, are shore-mounted and are quiet. Learn more about our nanobubbles generators on our product page.

Implement an integrated approach to lake management
ORP management should be part of a larger integrated approach to lake management that considers the various factors that can affect water quality. This may include addressing nutrient inputs, managing aquatic vegetation, and controlling invasive species.
By following these best practices, lake and pond managers can help maintain healthy water quality conditions and promote a sustainable aquatic ecosystem.


